Mass Spectrometry Matrix Effect Study for the Trace Analysis of Chloramphenicol Residues in Honey
-
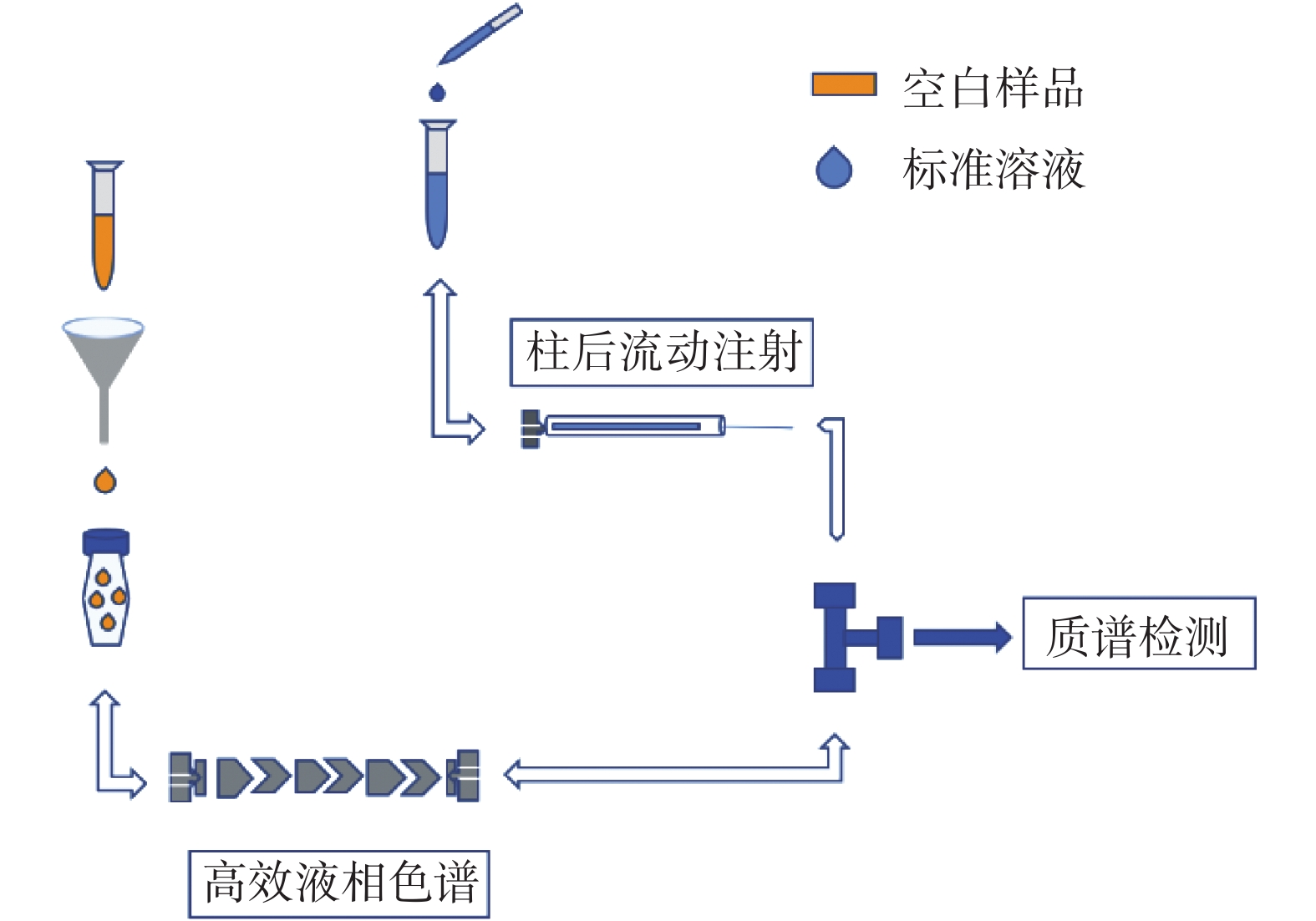
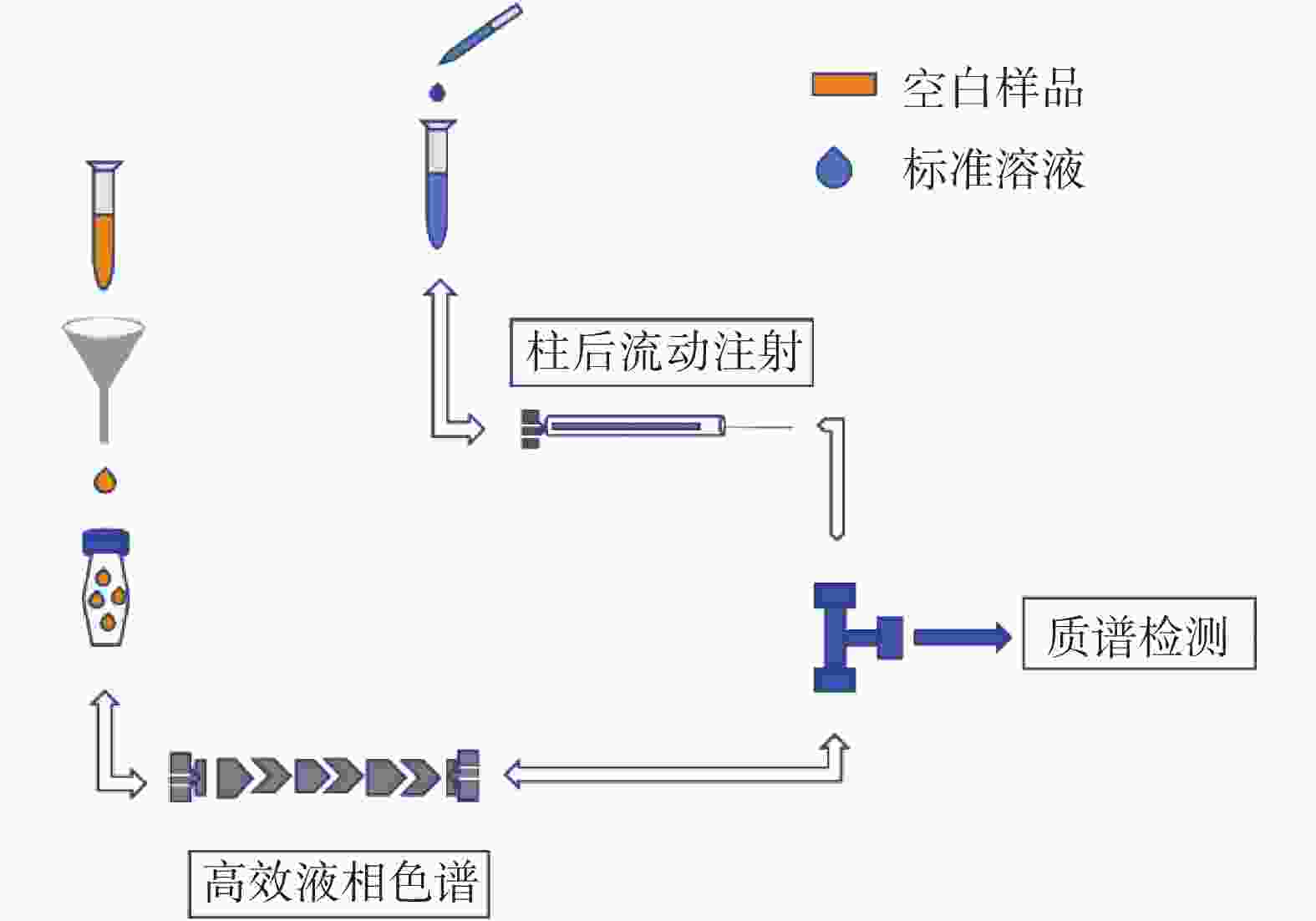
摘要: 有机同位素稀释质谱法(Isotope dilution mass spectrometry,IDMS)是复杂基质样品中痕量成分准确测量的潜在基准方法,但分析条件、同位素稀释试剂、基质效应(Matrix Effect,ME))等因素也会对结果产生影响。本文以枣花和荆花蜂蜜为研究样品,比较不同提取方法下蜂蜜中氯霉素痕量残留测量时的基质效应,研究基质效应对结果的影响、基质效应的补偿及消除方法。采用基质效应柱后注射定性评价方法和萃取后添加定量评价方法,考察直接稀释提取法和乙酸乙酯提取法的基质效应及其对氯霉素测量结果准确性的影响;结果表明,直接稀释提取溶液中氯霉素受到的基质抑制效应更为显著,ME在7%~10%之间,抑制了90%以上的氯霉素质谱响应,外标法校准的提取回收率仅为10.9%±1.9%;乙酸乙酯法提取溶液的ME在33%~35%之间,基体抑制相对较弱,外标法校准的提取回收率为33.7%±1.2%。采用同位素稀释质谱法进行校准,两种提取方法的回收率分别为100.4%±4.3%、100.2%±3.4%,说明基质抑制效应对测量结果的影响被有效补偿。直接稀释提取法采用IDMS校准,可准确、快速测量蜂蜜中氯霉素痕量残留。
-
关键词:
- 蜂蜜 /
- 氯霉素 /
- 基质效应 /
- 同位素稀释质谱法 /
- 液相色谱串联三重四级杆质谱(LC-MS/MS)
Abstract: Organic isotope dilution mass spectrometry (IDMS) is a potential reference method for the accurate measurement of trace components in complex matrix samples. However, the analytical conditions, isotope dilution reagents, matrix effects (MEs), and other factors can also affect the results. In this paper, jujube and wattle honey were used as the study samples to compare the MEs in the trace measurement of chloramphenicol residues in honey under different extraction methods, so as to investigate the influence of MEs on the results, the compensation and elimination methods of MEs. -
表 1 LC-MS/MS检测离子对信息
Table 1. Transition reactions monitored by LC- MS/MS
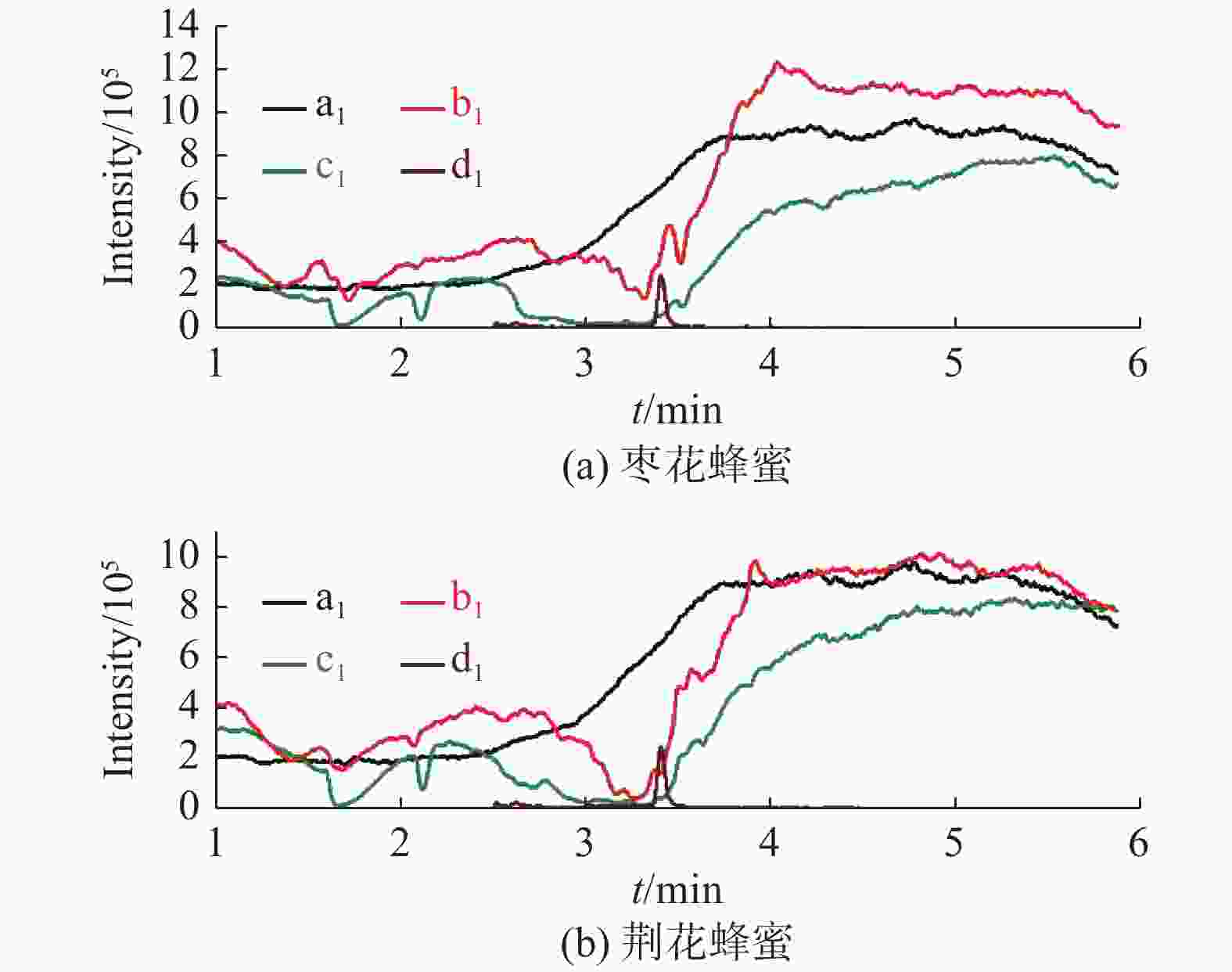
分析物 保留时间/min 母离子(m/z) 子离子(m/z) 碰撞电压/V CAP 3.4 321 152* −17 194 −11 257 −10 D5-CAP 3.4 326 157* −17 199 −11 262 −10 *定量离子 表 2 直接稀释法与乙酸乙酯提取法应用于不同蜂蜜基质中的ME与θ值(n=3)
Table 2. ME and correction factor of direct dilution method and ethyl acetate extraction method applied to different honey(n =3)
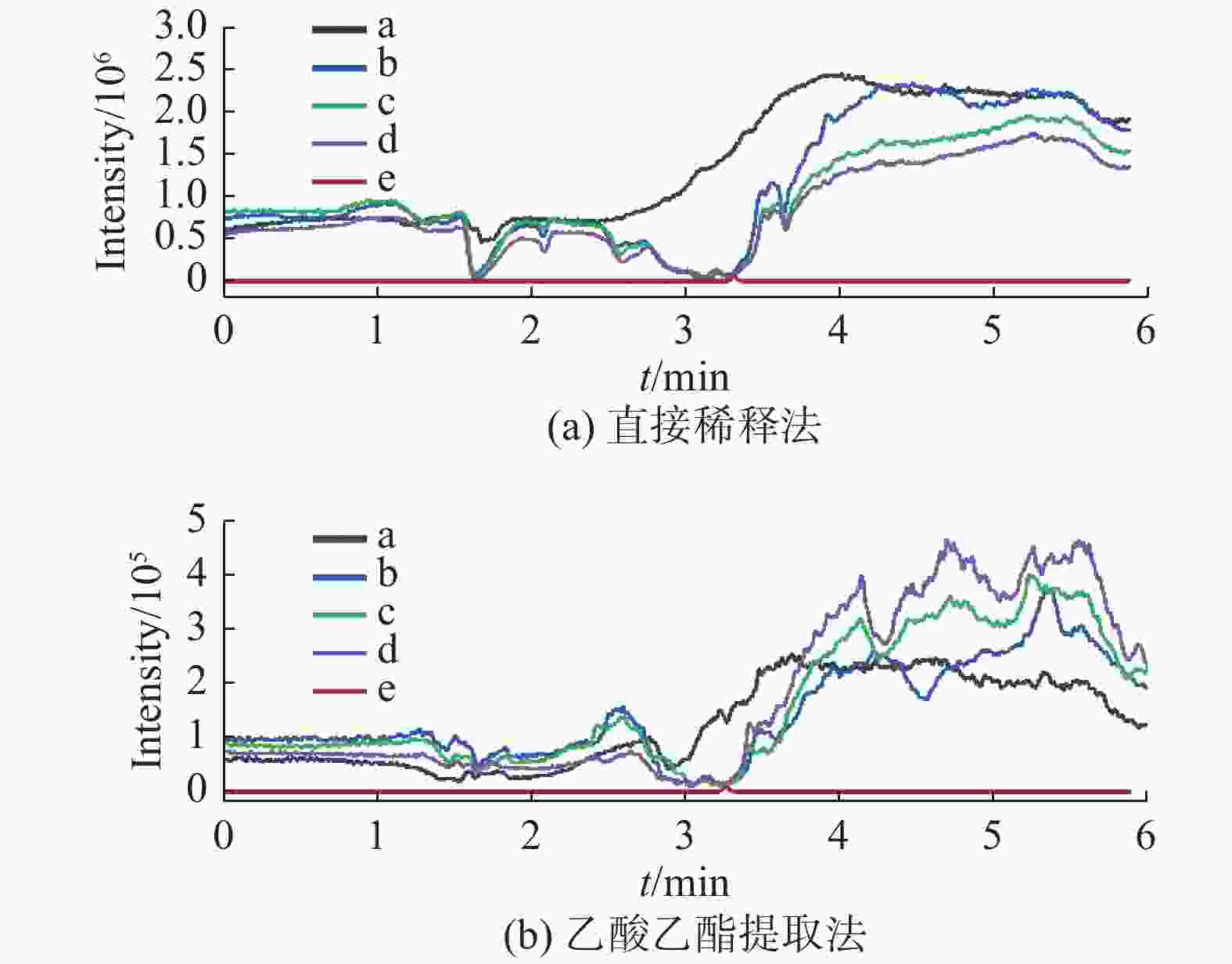
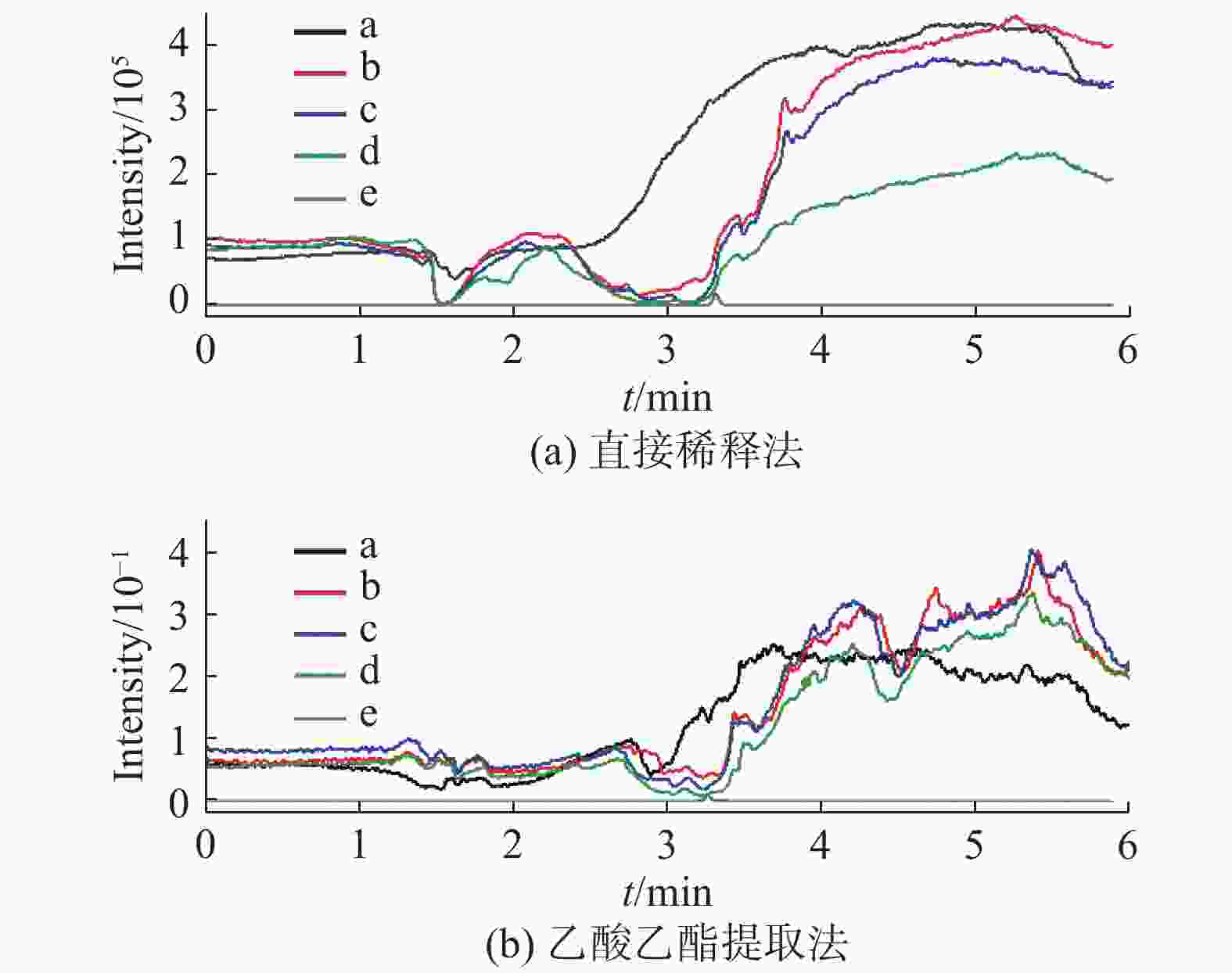
基质效应 枣花蜂蜜 荆花蜂蜜 直接稀释法 MEcap/% 10.4 ± 0.7 7.6 ± 1.0 MEd5-cap/% 10.2 ± 0.9 7.6 ± 1.1 θ 1.03 ± 0.03 1.01 ± 0.04 乙酸乙酯提取法 MEcap/% 35.2 ± 2.6 33.3 ± 4.0 MEd5-cap/% 36.3 ± 2.5 34.5 ± 3.8 θ 0.97 ± 0.02 0.97 ± 0.03 表 3 两种提取溶液不同净化条件的基质效应(n=6)
Table 3. Matrix effect optimized by two extraction methods (n = 6)
优化条件 直接稀释法 乙酸乙酯提取法 MEcap/% MEd5-cap/% MEcap/% MEd5-cap/% 水体积/mL 20 11.2 ± 0.6 11.6 ± 0.8 11.8 ± 1.0 11.5 ± 0.7 10 12.4 ± 0.9 12.8 ± 1.1 12.2 ± 1.5 12.3 ± 1.5 5 12.0 ± 1.0 12.0 ± 1.1 12.3 ± 1.0 12.2 ± 0.7 甲醇比例/% 50 11.9 ± 0.7 12.3 ± 0.7 21.5 ± 2.2 21.1 ± 1.9 25 6.5 ± 0.8 6.6 ± 0.8 13.2 ± 1.7 12.5 ± 1.9 5 4.1 ± 0.7 4.5 ± 1.1 12.5 ± 0.9 12.1 ± 1.0 表 4 直接稀释法和乙酸乙酯提取法提取枣花蜂蜜中的CAP回收率%(n=6)
Table 4. Recovery% of CAP in jujube honey by direct dilution method and ethyl acetate extraction method (n = 6)
浓度/μg·kg1 直接稀释法 乙酸乙酯提取法 RE1 RE2 RE3 RE4 RE1 RE2 RE3 RE4 0.1 10.7 ± 2.4 99.1 ± 4.8 101.8 ± 4.6 100.1 ± 4.9 31.3 ± 1.0 69.8 ± 5.2 101.1 ± 3.1 103.8 ± 4.7 0.4 11.3 ± 1.4 103.1 ± 3.2 102.2 ± 5.1 100.7 ± 4.4 30.0 ± 1.8 75.8 ± 4.3 97.8 ± 3.3 99.9 ± 2.8 2.0 10.8 ± 1.8 100.1 ± 2.5 100.6 ± 2.2 100.5 ± 3.8 33.8 ± 0.8 87.0 ± 3.0 97.6 ± 4.4 96.8 ± 2.9 平均回收率% 10.9 ± 1.9 100.8 ± 3.5 101.3 ± 3.9 100.4 ± 4.3 33.7 ± 1.2 77.6 ± 4.2 98.8 ± 3.6 100.2 ± 3.4 注:1.溶液外标校准法;2.基质匹配外标校准法;3. IDMS溶液校准法;4. IDMS基质匹配校准法 表 5 两种提取方法应用于不同实际蜂蜜样本中CAP的检测浓度μg·kg−1(n=5)
Table 5. Detection concentration of CAP in different real honey samples by two extraction methods μg·kg−1 (n = 5)
蜂蜜 直接稀释法 乙酸乙酯提取法 荆花 0.0861±0.0032 0.0841±0.0049 枣花 0.0233±0.0019 0.0277±0.0024 枇杷 0.0619±0.0015 0.0561±0.0021 椴树 1.05±0.07 1.07±0.02 -
[1] Veach B, Mudalige T, Barens P, et al. Quantitative Screening Method for Erythromycin and Tylosin in Honey Using RapidFire Mass Spectrometry[J]. Journal of AOAC International, 2018, 101(3): 897-903. doi: 10.5740/jaoacint.17-0262 [2] European Legislation Regarding Antibiotics in Honey: An Overview[EB/OL]. [2021-03-18]. http://europroxima.com. [3] QIN X F, WANG Q Q, GENG L P, et al. A “Signal-On” Photoelectrochemical Aptasensor Based on Graphene Quantum Dots-Sensitized TiO2 Nanotube Arrays for Sensitive Detection of Chloramphenicol[J]. Talanta, 2019, 197(1): 28-35. [4] 农业部峰产品质量监督检验测试中心. 绿色食品蜂产品: NY/T752-2012[S]. 北京. 中国标准出版社, 2012: 12. [5] Barreto F, Ribeiro C, Barcellos Hoff R, et al. Determination of Chloramphenicol, Thiamphenicol, Florfenicol and Florfenicol Amine in Poultry, Swine, Bovine and Fish by Liquid Chromatography-Tandem Mass Spectrometry[J]. Journal of Chromatography A, 2016, 1449: 48-53. doi: 10.1016/j.chroma.2016.04.024 [6] Jakšić S, Ratajac R, Prica N, et al. Methods of Determination of Antibiotic Residues in Honey[J]. Journal of Analytical Chemistry, 2018, 73(4): 317-324. doi: 10.1134/S1061934818040044 [7] Matuszewsk B, Constanzer M, Chavez-Eng C. Strategies for the Assessment of Matrix Effect in Quantitative Bioanalytical Methods Based on HPLC-MS/MS[J]. Analytical Chemistry, 2013, 75(13): 3019-3030. [8] Gómez-Pérez M, Plaza-Bolaños P, Romero-González R, et al. Comprehensive Qualitative and Quantitative Determination of Pesticides and Veterinary Drugs in Honey Using Liquid Chromatography-Orbitrap High Resolution Mass Spectrometry[J]. Journal of Chromatography A, 2012, 1248: 130-138. doi: 10.1016/j.chroma.2012.05.088 [9] YAN C, ZHANG J, YAO L, et al. Aptamer-Mediated Colorimetric Method for Rapid and Sensitive Detection of Chloramphenicol in Food[J]. Food Chemistry, 2018, 260: 208-212. doi: 10.1016/j.foodchem.2018.04.014 [10] LI Y G, LIU X H, ZHANG R, et al. Analysis of Chloramphenicol in Drinking Water Using an Evaporation Preparative Step and Isotope Dilution Liquid Ehromatography-Tandem Mass Spectrometry[J]. Acta Chromatographica, 2018, 30(1): 17-20. doi: 10.1556/1326.2017.28404 [11] Galarini R, Saluti G, Giusepponi D, et al. Multiclass determination of 27 Antibiotics in Honey[J]. Food Control, 2015, 48: 12-24. doi: 10.1016/j.foodcont.2014.03.048 [12] LI Z W, LEI C, WANG N, et al. Preparation of Magnetic Molecularly Imprinted Polymers with Double Functional Monomers for the Extraction and Detection of Chloramphenicol in Food[J]. Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences, 2018, 1100-1101(9): 113-121. [13] Armenta S, de la Guardia M, Abad-Fuentes A, et al. Highly Selective Solid-Phase Extraction Sorbents for Chloramphenicol Determination in Food and Urine by Ion Mobility Spectrometry[J]. Analytical and Bioanalytical Chemistry, 2016, 408(29): 8559-8567. doi: 10.1007/s00216-016-9995-9 [14] Sniegocki T, Posyniak A, Gbylik-Sikorska M, et al. Determination of Chloramphenicol in Milk Using a QuEChERS-Based on Liquid Chromatography Tandem Mass Spectrometry Method[J]. Analytical Letters, 2014, 474(4): 568-578. [15] Rezaee M, Khalilian F. Application of Ultrasound-Assisted Extraction Followed by Solid-Phase Extraction Followed by Dispersive Liquid-Liquid Microextraction for the Determination of Chloramphenicol in Chicken Meat[J]. Food Analytical Methods, 2018, 11(3): 759-767. doi: 10.1007/s12161-017-1048-2 [16] Baeza Fonte A, Rodríguez Castro G, Liva-Garrido M. Multi-Residue Analysis of Sulfonamide Antibiotics in Honey Samples by On-line Solid Phase Extraction Using Molecularly Imprinted Polymers Coupled to Liquid Chromatography-Tandem Mass Spectrometry[J]. Journal of Liquid Chromatography and Related Technologies, 2018, 41(15-16): 881-891. doi: 10.1080/10826076.2018.1533477 [17] Kawano S, HAO HongYuan, Hashi Y, et al. Analysis of Chloramphenicol in Honey by On-line Pretreatment Liquid Chromatography-Tandem Mass Spectrometry[J]. Chinese Chemical Letters, 2015, 26(1): 36-38. doi: 10.1016/j.cclet.2014.10.026 [18] Shao B, Jia X, Zhang J, et al. Multi-residual analysis of 16 β-agonists in pig liver, kidney and muscle by ultra performance liquid chromatography tandem mass spectrometry[J]. Food Chem, 2009, 114(11): 15-21. [19] Berg T, Karlsen M, Øiestad Å M L, et al. Evaluation of 13C- and 2H-labeled internal standards for the determination of amphetamines in biological samples, by reversed-phase ultra-high performance liquid chromatography-tandem mass spectrometry[J]. Journal of Chromatography A, 2014, 1344: 83-90. doi: 10.1016/j.chroma.2014.04.020 [20] Berg T, Strand D H. 13C labelled internal standards-A solution to minimize ion suppression effects in liquid chromatography-tandem mass spectrometry analyses of drugs in biological samples[J]. Journal of Chromatography A, 2011, 1218(52): 9366-9374. doi: 10.1016/j.chroma.2011.10.081 [21] Cappiello A, Famiglini G, Palma P, et al. Overcoming Matrix Effects in Liquid Chromatography-Mass Spectrometry[J]. Analytical Chemistry, 2008, 80(23): 9343-9348. doi: 10.1021/ac8018312 [22] Bienvenu J, Provencher G, Bélanger P, et al. Standardized Procedure for the Simultaneous Determination of the Matrix Effect, Recovery, Process Efficiency, and Internal Standard Association[J]. Analytical Chemistry, 2017, 89(14): 7560-7568. doi: 10.1021/acs.analchem.7b01383 [23] LI X Q, LI H M, XU S, et al. Rapid Quantification of Trace Chloramphenicol in Honey Under Ambient Conditions Using Direct Analysis Via Real-Time QTRAP Mass Spectrometry[J]. Food Chemistry, 2019, 276: 50-56. doi: 10.1016/j.foodchem.2018.09.130 [24] LI X Q, YANG Z, ZHANG Q H, et al. Evaluation of Matrix Effect in Isotope Dilution Mass Spectrometry Dased on Quantitative Analysis of Chloramphenicol Residues in Milk Powder[J]. Analytica Chimica Acta, 2014, 807: 75-83. doi: 10.1016/j.aca.2013.11.017 [25] Sargent M, Harte R, Harrington C. Guidelines for Achieving High Accuracy in Isotope Dilution Mass Spectrometry (IDMS)[M]. Laboratory of the Government Chemist by The Royal Society of Chemistry, 2002. [26] Mottier P, Parisod V, Gremaud E, et al. Determination of the Antibiotic Chloramphenicol in Meat and Seafood Products by Liquid Chromatography-Electrospray Ionization Tandem Mass Spectrometry[J]. Journal of Chromatography A, 2003, 994(1-2): 75-84. doi: 10.1016/S0021-9673(03)00484-9 [27] Lopez M, Pettis J, Smith I, et al. Multiclass Determination and Confirmation of Antibiotic Residues in Honey Using LC-MS/MS[J]. Journal of Agricultural and Food Chemistry, 2008, 56(5): 1553-1559. doi: 10.1021/jf073236w [28] Pagliano E, Meija J. Reducing the Matrix Effects in Chemical Analysis: Fusion of Isotope Dilution and Standard Addition Methods[J]. Metrologia, 2016, 53(2): 829-834. doi: 10.1088/0026-1394/53/2/829 -

 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公

 下载:
下载: