Preparation of Rimantadine Hydrochloride Purity Certified Reference Material
-
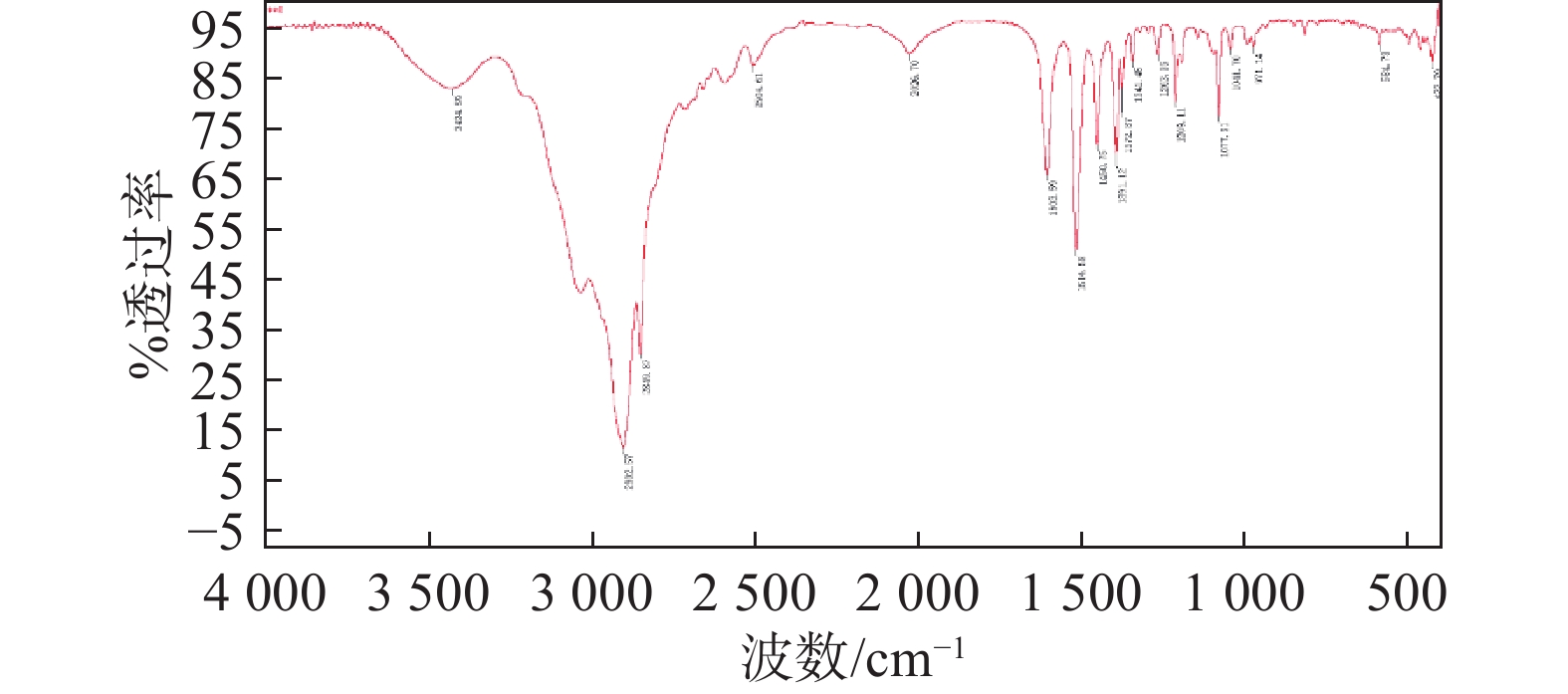
摘要: 采用“质量平衡法”和“定量核磁法”两种独立方法研制了盐酸金刚乙胺纯度标准物质(GBW10262)。对标准物质研制过程中的关键因素,如均匀性、稳定性、定值和不确定度等进行了系统研究。两种方法测得的盐酸金刚乙胺纯度值为99.7%±0.3%(k=2)。所研制的盐酸金刚乙胺标准物质均匀性良好,长期稳定性可保持20个月,并且在7天内保持稳定(50 °C)。同时,对定值、稳定性、均匀性等不确定度来源进行了评估。所研制的盐酸金刚乙胺有证标准物质将为仪器检定、方法验证、质量安全风险监测等实施提供支撑。Abstract: The development of a rimantadine hydrochloride purity certified reference material (CRM) (GBW 10262) was carried out using two independent methods: the mass balance (MB) method and quantitative nuclear magnetic resonance (qNMR) spectroscopy. Critical aspects of the CRM development process, including homogeneity, stability, calibration, and uncertainty, were systematically investigated. The purity of rimantadine hydrochloride CRM, as determined by both methods, was found to be 99.7%±0.3% (k=2). The CRM demonstrated excellent homogeneity, maintaining long-term stability for 20 months, and short-term stability for seven days at 50°C. The study also involved a thorough assessment of uncertainty sources, including characterization, stability, and homogeneity. The rimantadine hydrochloride CRM will support activities such as instrument calibration, method validation, and quality and safety risk monitoring.
-
Key words:
- metrology /
- rimantadine /
- homogeneity /
- purity /
- certified reference material /
- uncertainty evaluation /
- MB /
- qNMR
-
表 1 盐酸金刚乙胺中水分含量(%)
Table 1. Percentage of moisture content in rimantadine hydrochloride
样品 水分含量 平均值 标准偏差 1 0.11 0.13 0.022 2 0.17 3 0.13 4 0.15 5 0.12 6 0.16 7 0.11 表 2 盐酸金刚乙胺中氯离子摩尔含量(mmol)
Table 2. Molar content of chloride ions in rimantadine hydrochloride
样品 氯离子摩尔含量 平均值 标准偏差 1 0.004802 0.004611 0.000214 2 0.004385 3 0.004723 4 0.004743 5 0.004858 6 0.004520 7 0.004246 表 3 盐酸金刚乙胺的质量平衡法定值结果(%)
Table 3. Purity determination of rimantadine hydrochloride CRM using the mass balance method
样品 有机纯度 水分含量 挥发性组分 不挥发性组分 定值结果 1 99.96 0.11 0.0146 0.0148 99.82 2 99.92 0.17 99.72 3 99.97 0.13 99.81 4 99.90 0.15 99.72 5 99.97 0.12 99.82 6 99.89 0.16 99.70 7 99.85 0.11 99.71 平均值 99.92 0.13 0.0146 0.0148 99.76 标准偏差 0.043 0.022 - - 0.0520 表 4 盐酸金刚乙胺的定量核磁法定值结果(%)
Table 4. Purity determination of rimantadine hydrochloride CRM using the qNMR method
样品 $ \dfrac{{I}_{\mathrm{x}}}{{I}_{\mathrm{s}\mathrm{t}\mathrm{d}}} $ $ \dfrac{{N}_{\mathrm{s}\mathrm{t}\mathrm{d}}}{{N}_{\mathrm{x}}} $ $ \dfrac{{M}_{\mathrm{x}}}{{M}_{\mathrm{s}\mathrm{t}\mathrm{d}}} $ $ \dfrac{{m}_{\mathrm{s}\mathrm{t}\mathrm{d}}}{{m}_{\mathrm{x}}} $ $ P_{\mathrm{q}\mathrm{N}\mathrm{M}\mathrm{R}\mathrm{ }} $ 1 0.5652 2 0.8726 1.0121 99.82 2 0.5883 2 0.8726 0.9726 99.84 3 0.5866 2 0.8726 0.9747 99.78 4 0.5699 2 0.8726 1.0015 99.59 5 0.5652 2 0.8726 1.0102 99.64 6 0.5758 2 0.8726 0.9926 99.74 7 0.5765 2 0.8726 0.9906 99.65 平均值 99.72% 标准偏差 0.10% 表 5 盐酸金刚乙胺不确定度评估(%)
Table 5. Uncertainty evaluation for rimantadine hydrochloride CRM
不确定度来源 计算公式 不确定度 均匀性 $ \dfrac{\sqrt{\dfrac{{{s}_{2}}^{2}}{n}}*\sqrt[4]{\dfrac{2}{{v}_{2}}}}{\stackrel{-}{X}} $ 0.0107 长期稳定性 $ \dfrac{\mathrm{s}\left({\beta }_{1}\right)\times t}{\stackrel{-}{X}} $ 0.021 短期稳定性 $ \dfrac{\mathrm{s}\left({\beta }_{1}\right)\times t}{\stackrel{-}{X}} $ 0.011 质量平衡法 $ \dfrac{{P}_{{\mathrm{HPLC-AN}}}.\sqrt{{\left(\dfrac{u\left({P}_{0}\right)}{{P}_{0}}\right)}^{2}+\dfrac{{u}^{2}\left({X}_{w}\right)+{u}^{2}\left({X}_{n}\right)+{u}^{2}\left({X}_{v}\right)}{{(1-{X}_{w}-{X}_{n}-{X}_{v})}^{2}}}}{\stackrel{-}{X}} $ 0.138 定量核磁法 $ \sqrt{{\left(\dfrac{u\left({I}_{\mathrm{x}}/{I}_{\mathrm{s}\mathrm{t}\mathrm{d}}\right)}{{I}_{\mathrm{x}}/{I}_{\mathrm{s}\mathrm{t}\mathrm{d}}}\right)}^{2}+{\left(\dfrac{u\left({M}_{\mathrm{x}}\right)}{{M}_{\mathrm{x}}}\right)}^{2}+{\left(\dfrac{u\left({M}_{\mathrm{s}\mathrm{t}\mathrm{d}}\right)}{{M}_{\mathrm{s}\mathrm{t}\mathrm{d}}}\right)}^{2}+{\left(\dfrac{u\left({m}_{\mathrm{x}}\right)}{{m}_{\mathrm{x}}}\right)}^{2}+{\left(\dfrac{u\left({m}_{\mathrm{s}\mathrm{t}\mathrm{d}}\right)}{{m}_{\mathrm{s}\mathrm{t}\mathrm{d}}}\right)}^{2}+{\left(\dfrac{u\left({p}_{\mathrm{s}\mathrm{t}\mathrm{d}}\right)}{{p}_{\mathrm{s}\mathrm{t}\mathrm{d}}}\right)}^{2}} $ 0.151 定值 $ \sqrt{\left[\dfrac{u_{\mathrm{r}\mathrm{e}\mathrm{l}}\left(P_{\mathrm{M}\mathrm{B}}\right)}{2}\right]^2+\left[\dfrac{u_{\mathrm{r}\mathrm{e}\mathrm{l}}\left(P_{\mathrm{q}\mathrm{N}\mathrm{M}\mathrm{R}}\right)}{2}\right]^2} $ 0.102 相对不确定度 $ \sqrt{u_{\mathrm{char},\mathrm{r}\mathrm{e}\mathrm{l}}^2+u_{\mathrm{bb},\mathrm{r}\mathrm{e}\mathrm{l}}^2+u_{\mathrm{lts},\mathrm{r}\mathrm{e}\mathrm{l}}^2+u_{\mathrm{sts},\mathrm{r}\mathrm{e}\mathrm{l}}^2} $ 0.106 包含因子 k 2 扩展不确定度 $ k\times u\mathrm{_{CRM}} $ 0.3 -
[1] Dolin R, Reichman R C, Madore H P, et al. A controlled trial of amantadine and rimantadine in the prophylaxis of influenza a infection[J]. NEW ENGLAND JOURNAL OF MEDICINE, 1982, 307: 580-584. doi: 10.1056/NEJM198209023071002 [2] 张美, 朱莉莎, 徐飘飘, 等. 高效液相色谱-串联质谱法同时检测鸡蛋中金刚乙胺和金刚烷胺药物残留研究[J]. 中国家禽, 2023, 45(2): 71-76. doi: 10.16372/j.issn.1004-6364.2023.02.012 [3] Hubsher G, Haider M, Okun M S. Amantadine: The journey from fighting flu to treating Parkinson disease[J]. NEUROLOGY, 2012, 78: 1096-1099. doi: 10.1212/WNL.0b013e31824e8f0d [4] 中华人民共和国农业部公告第560号[Z]. http://www.moa.gov.cn/nybgb/2005/dsyq/201806/t20180618_6152552.htm. [5] 陆淳, 张鑫, 吴剑平, 等. 抗病毒药物在动物组织中残留检测方法进展[J]. 中国兽医杂志, 2016, 52(6): 66-68. doi: 10.3969/j.issn.0529-6005.2016.06.014 [6] 赵军强, 韩典峰, 田秀慧, 等. 食品中金刚烷胺的危害、检测方法和残留风险研究进展[J]. 中国渔业质量与标准, 2022, 12(3): 64-71. doi: 10.3969/j.issn.2095-1833.2022.03.010 [7] 孙玲玲, 霍江莲, 陈冬东, 等. 液相色谱-串联质谱法同时测定鸡蛋中4种抗病毒药物残留[J]. 质量安全与检验检测, 2023, 33(5): 6-13,30. [8] 王小乔, 李坚, 许晓辉, 等. QuEChERS-UPLC-MS/MS测定鱼肉中3种抗病毒药物残留[J]. 中国兽药杂志, 2023, 57(8): 35-42. [9] 欧阳思, 雨蓝伟, 贾佳, 等. 超高效液相色谱-串联质谱法同时测定鸡肉、鸡蛋中尼卡巴嗪、金刚烷胺、金刚乙胺残留量[J]. 现代食品, 2022, 28(23): 138-141,147. doi: 10.16736/j.cnki.cn41-1434/ts.2022.23.037 [10] 张季, 凌蕾, 卢叶, 等. 同位素内标-UPLC-MS/MS法同时测定羊肉中金刚烷胺和金刚乙胺的残留量[J]. 广州化工, 2022, 50(4): 106-108. doi: 10.3969/j.issn.1001-9677.2022.04.035 [11] Zhang T, Zhang L, Liu J X, et al. Development of a molecularly imprinted microspheres-based microplate fluorescence method for detection of amantadine and rimantadine in chicken[J]. FOOD ADDITIVES AND CONTAMINANTS PART A-CHEMISTRY ANALYSIS CONTROL EXPOSURE & RISK ASSESSMENT, 2021, 38(7): 1136-1147. [12] Nuchtavorn N, Sudprasert C, Yurai P, et al. A simple and rapid microplate fluorescence determination of adamantanes in pharmaceutical formulations[J]. CHEMICAL PAPERS, 2020, 74: 1859-1865. doi: 10.1007/s11696-019-01035-x [13] Wang Z, Wang X, Wang Y, et al. Simultaneous determination of five antiviral drug residues and stability studies in honey using a two-step fraction capture coupled to liquid chromatography tandem mass spectrometry[J]. JOURNAL OF CHROMATOGRAPHY A, 2021, 1638: 461890. doi: 10.1016/j.chroma.2021.461890 [14] Sidiropoulou G, Kabir A, Furton K G, et al. Combination of fabric phase sorptive extraction with UHPLC-ESI-MS/MS for the determination of adamantine analogues in human urine[J]. MICROCHEMICAL JOURNAL, 2022, 176(10): 107250. [15] ISO. Reference materials-selected terms and definitions : Guide ISO. 30[S]. ISO: Geneva, 2015. [16] 于亚东, 刘媛. ISO/REMCO关于标准物质的新定义[J]. 化学分析计量, 2008, 2: 64-65. doi: 10.3969/j.issn.1008-6145.2008.02.021 [17] 国家市场监督管理总局. 标准物质的定值及稳定性、均匀性评估: JJF 1343-2022 [S]. 北京: 中国标准出版社, 2022. [18] Wang S, Wu P, Li M, et al. Mass balance method for SI-traceable purity assignment of synthetic oxytocin[J]. JOURNAL OF PHARMACEUTICAL AND BIOMEDICAL ANALYSIS, 2022, 207: 114401. doi: 10.1016/j.jpba.2021.114401 [19] Wang S, Wang S, Li P, et al. Establishment of SI-traceable purity assessment of Fumonisin B1 using a combination of quantitative 1H NMR and mass balance[J]. MICROCHEMICAL JOURNAL, 2023, 185: 108282. doi: 10.1016/j.microc.2022.108282 [20] Westwood S, Choteau T, Daireaux A, et al. Mass Balance Method for the SI Value Assignment of the Purity of Organic Compounds[J]. ANALYTICAL CHEMISTRY, 2013, 85: 3118-3126. doi: 10.1021/ac303329k [21] Liu H, Cheow P S, Yong S, et al. Determination of purity values of amino acid reference materials by mass balance method: An approach to the quantification of related structure impurities[J]. ANALYTICAL AND BIOANALYTICAL CHEMISTRY, 2020, 412: 8023-8037. doi: 10.1007/s00216-020-02936-7 [22] Guo Q, Li Z, Shen L, et al. Quantitative 1H nuclear magnetic resonance (qHNMR) methods for accurate purity determination of glucosinolates isolated from Isatis indigotica roots[J]. PHYTOCHEMICAL ANALYSIS, 2021, 32: 104-111. doi: 10.1002/pca.3003 [23] Zhou J, Li F, Wang M, et al. Preparation of clorprenaline certified reference material: Purity determination and uncertainty evaluation[J]. MICROCHEMICAL JOURNAL, 2022, 179: 107502. doi: 10.1016/j.microc.2022.107502 [24] Chen W, Jin W, Fang H, et al. Development of a new taurine purity certified reference material[J]. MICROCHEMICAL JOURNAL, 2022, 181: 107761. doi: 10.1016/j.microc.2022.107761 [25] Zhang L, Yuan P H, Yang D Z, et al. Purity and uncertainty study of CRM Betulin by DSC[J]. NATURAL PRODUCTS AND BIOPROSPECTING, 2020, 10: 317-324. doi: 10.1007/s13659-020-00261-1 [26] Bruni G, Berbenni V, Milanese C, et al. Determination of the nateglinide polymorphic purity through DSC[J]. JOURNAL OF PHARMACEUTICAL AND BIOMEDICAL ANALYSIS, 2011, 54: 1196-1199. doi: 10.1016/j.jpba.2010.12.003 [27] 苏斌, 张宝喜, 毕研才, 等. 咖啡酸国家一级纯度标准物质的研制[J]. 医药导报, 2023, 42(8): 1192-1198. [28] 国家质量监督检验检疫总局. 一级标准物质技术规范: JJF 1006-1994 [S]. 北京: 中国计量出版社, 1994. [29] Quan C. Establishment of the purity values of carbohydrate certified reference materials using quantitative nuclear magnetic resonance and mass balance approach[J]. FOOD CHEMISTRY, 2014, 153: 378-386. doi: 10.1016/j.foodchem.2013.12.086 [30] 国家质量监督检验检疫总局. 标准物质研制生产机构通用技术要求: JJF 1342-2012 [S]. 北京: 中国质检出版社, 2012. -

 作者投稿
作者投稿 专家审稿
专家审稿 编辑办公
编辑办公


 下载:
下载: